

Bionnovation Surgitime Titanium
$123.87 – $1,165.82 inc. GST
Bionnovation Surgitime Titanium provides excellent biocompatibility, occlusive property, its permeability enables the transmission of nutrients.
Bionnovation Surgitime Titanium
Bionnovation Surgitime Titanium (Titanium Mesh) is a non-absorbable titanium screen made with pure Titanium (ASTM F-67). It comes in different sizes, thicknesses and hole diameters in order to meet varying clinical needs.
Product is supplied sterile. Must be kept under ideal storage and preservation conditions and the integrity of the pack maintained.
- Gamma Radiation (25kGy).
Purpose
Surgitime Titanium aids in bone neoformation, acting as a barrier that prevents the migration of epithelial cells and connective tissue, avoiding the competition with the bone graft.
Benefits
- Excellent biocompatibility
- Occlusive property
- Permeability enables the transmission of nutrients
- Easy use because it is very malleable and can be cut to adapt to surgical sites
- Has the capacity of keeping the regenerative space whole
- Enables vascularization of the graft on both sides (periosteum and endosteum).
Designed to ensure the three-dimensional reconstruction of alveolar bone defects and to facilitate bone replacement through suitable fixation of the replacement material.
Consideration
Immobilization of the graft / membrane complex is a major factor for the success of bone reconstruction. It also depends on the correct selection of grafting biomaterials and the use of regenerative membranes. Care with the size and location of the flap, careful removal without damaging the periosteum and primary closure without tension, are fundamental for good postoperative outcomes.
Surgitime Titanium should be shaped according to the anatomy of the bone and should not be bent at sharp angles, scratched or deformed. Once used and molded, it should not be molded again as it may result in product function failure.
Must stay in situ for at least 21 days for osteoconduction to start.
Advantages
- Easy to use in surgical sites (Flexible)
- No trauma on soft tissues
- Suitable containment of the bone graft
- Improves the space for bone regeneration
- Ultra fine (0.04 mm and 0.08 mm)
- Biocompatible
- Pure Grade 1 Titanium
| Brand |
Bionnovation |
|---|---|
| Options |
Surgitime Titanium 34 x 25 mm (Thickness 0.04 mm – Hole 0.85 mm) ,Surgitime Titanium 34 x 25 mm (Thickness 0.04 mm – Hole 0.15 mm) ,Surgitime Titanium 34 x 25 mm (Thickness 0.08 mm – Hole 0.85 mm) ,Surgitime Titanium 60 x 60 mm (Thickness 0.08 mm – Hole 0.85 mm) ,Surgitime Titanium 120 x 120 mm (Thickness 0.08 mm – Hole 0.85 mm) ,Surgitime Titanium 200 x 200 mm (Thickness 0,08 mm – Hole 0,85 mm) |
Only logged in customers who have purchased this product may leave a review.
Related products
A-PRF Red Tubes
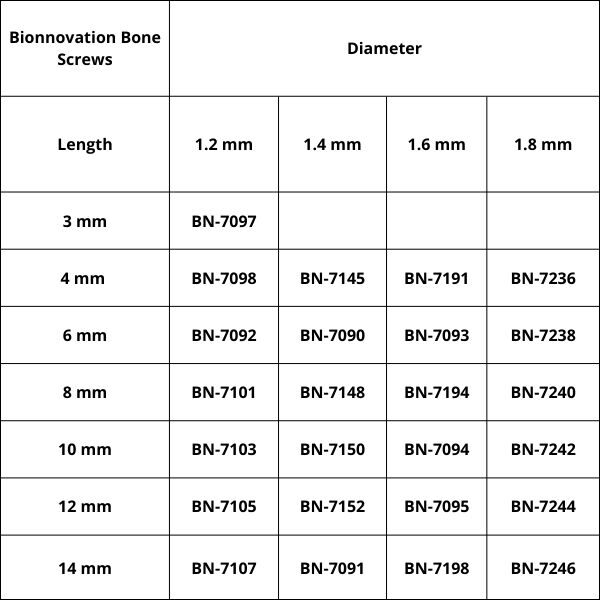
Bionnovation Bone Screws
Bionnovation Bone Grafting Screw Fixation Kit
- The screws are temporary, and they only remain within the bone repair period, as its purpose is to keep either the graft or the membrane in position and not for the purpose of osseo-integration
- Self-tapping
- With conic edge, cylindrical shaped body and cross-fit head and it is intended for the fixation of bone grafts and membranes
Bionnovation Cortical Drill
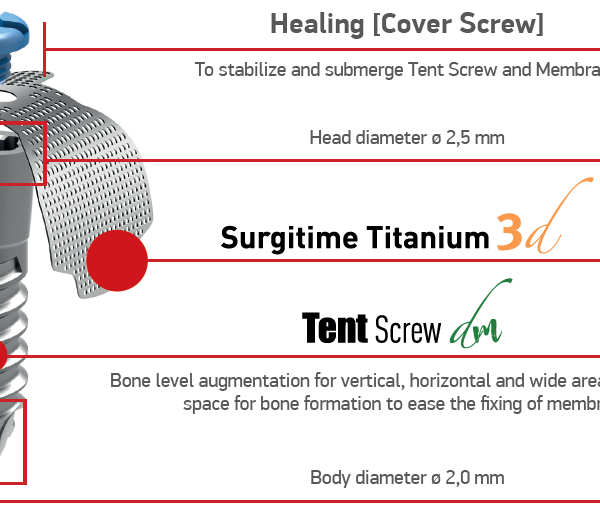
Bionnovation Tent Screw DM
A better answer for a successful GBR procedure.
Tent Screw DM is designed with a self-drilling tip, polished neck, and broader head to maintain space under resorbable and non-resorbable membranes in horizontal and vertical bone regeneration procedures.












Reviews
There are no reviews yet