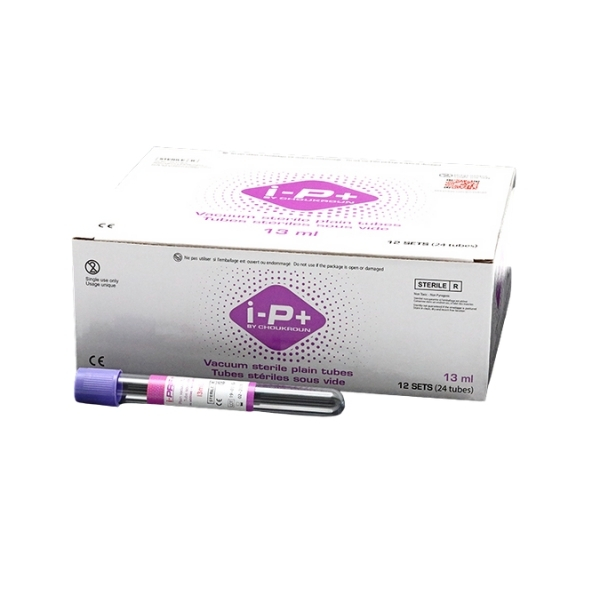
I-PRF Purple Tubes
I-PRF Purple Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Immediate Platelet-Rich Fibrin (I-PRF) from autologous blood. These tubes are designed for clinical use in regenerative medicine and aesthetic procedures.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
CE 0123
The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence
Expiry date: 04-2027
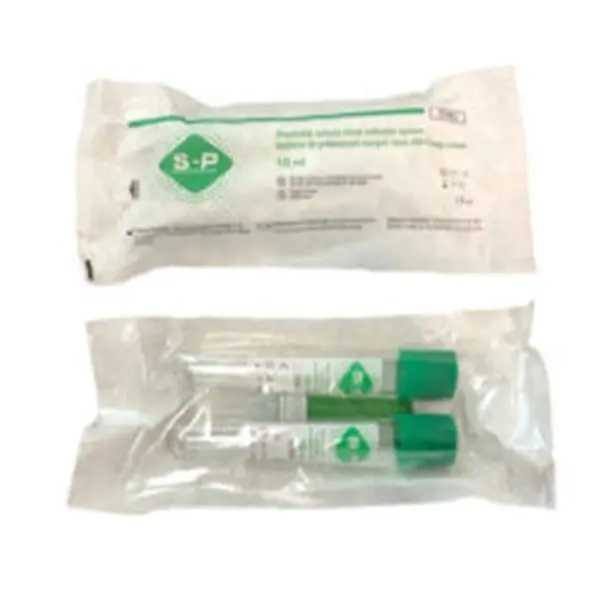
S-PRF Green Tubes
S-PRF Green Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Liquid Platelet-Rich Fibrin (S-PRF) from autologous blood in dental and medical procedures.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
CE 0123
The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF
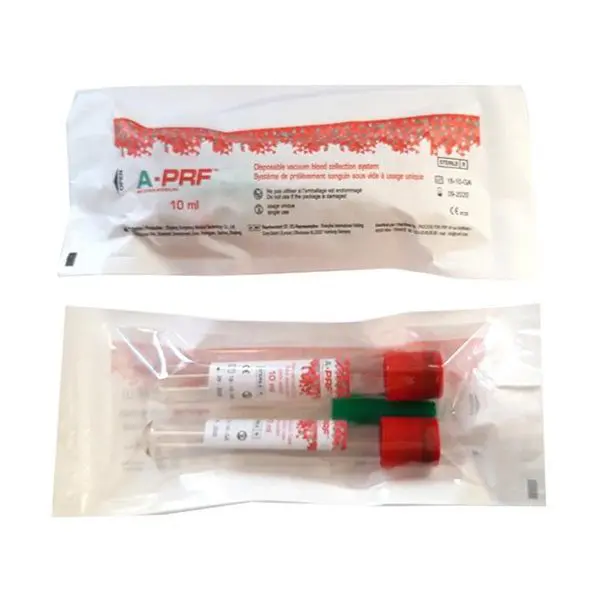
A-PRF Red Tubes
A-PRF Red Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Solid Platelet-Rich Fibrin (A-PRF) from autologous blood. They are specifically designed to facilitate the preparation of PRF clots and membranes for clinical use.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
CE 0123
The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence