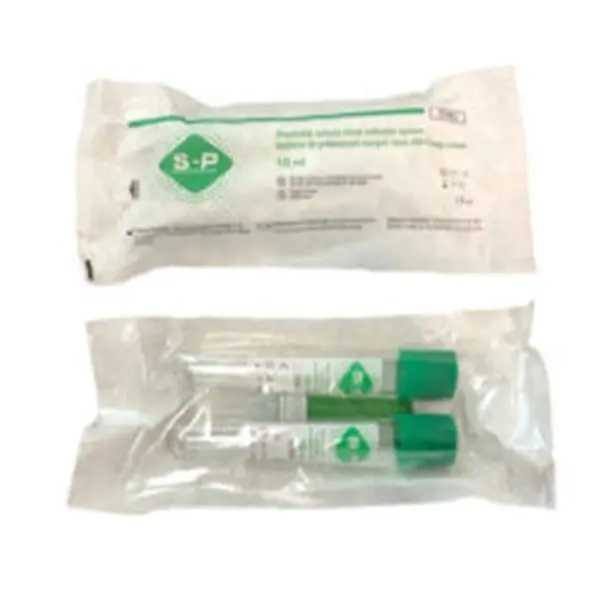
S-PRF Green Tubes
S-PRF Green Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Liquid Platelet-Rich Fibrin (S-PRF) from autologous blood in dental and medical procedures.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
CE 0123
The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF
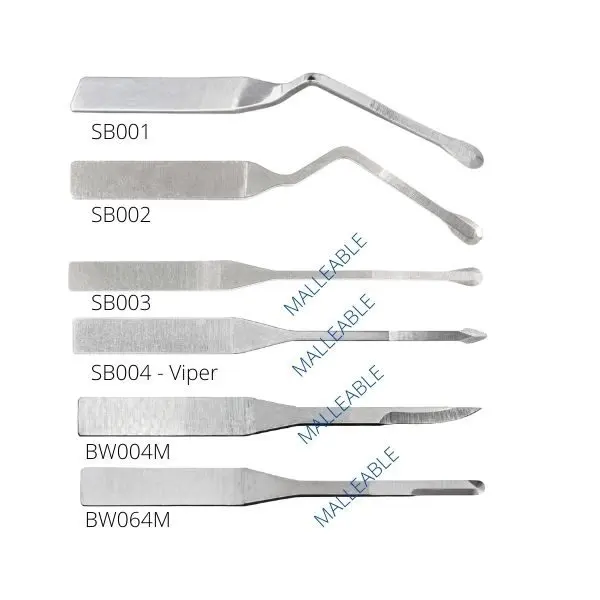
MJK Micro-blades – Periodontology
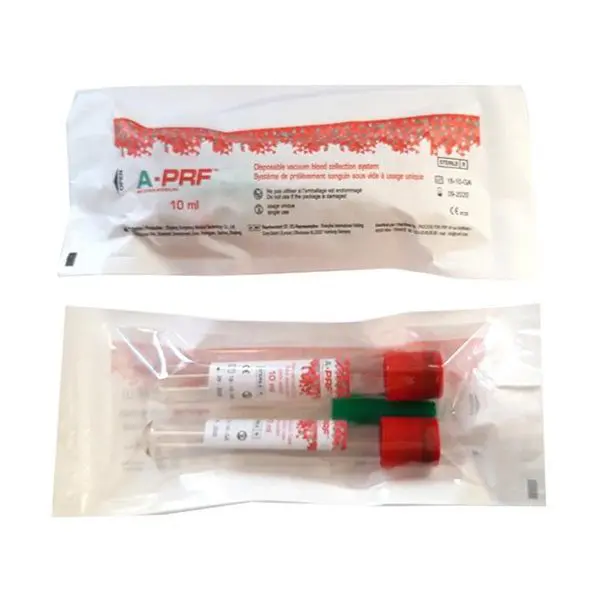
A-PRF Red Tubes
A-PRF Red Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Solid Platelet-Rich Fibrin (A-PRF) from autologous blood. They are specifically designed to facilitate the preparation of PRF clots and membranes for clinical use.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
CE 0123
The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence
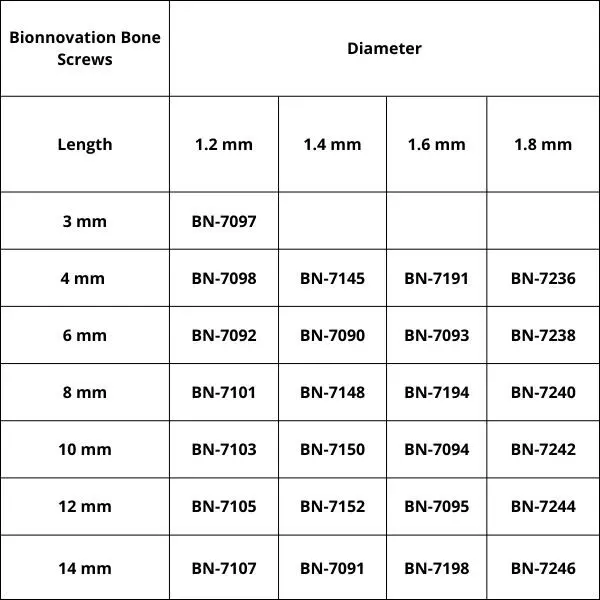
Bionnovation Bone Screws
Monoglyc Absorbable Monofilament Sutures
Monoglyc PGA-PCL Suture is a highly pliable monofilament suture, that also absorbs predictably. With the smooth monofilament surface structures, it handles and ties easily, and is virtually inert in tissue. Basically, PGA-PCL sutures behave like PTFE sutures, but do not need to be removed!
24 Sutures Per Box
Glycolon Suture
POMPAC from Process for PRF – Dr Joseph Choukroun
Brand: Process for PRF

The most important innovation in the field of PRF in the last 20 years!
In order to constantly improve the technique and results of PRF, discover our new product : PomPac™
With PomPac™ users will obtain larger PRF clots and liquid PRF with 50% more volume!
https://www.youtube.com/watch?v=0CoeS-p_Tyc
Bionnovation Surgitime Titanium SEAL
Brand: Bionnovation
Surgitime Titanium Seal (Titanium-Foil) is ideal for three-dimensional bone regeneration (GBR, Guided Bone Regeneration). If necessary, it can be attached with studs or screws.
Material
Titanium Grade I (ASTM F-67) : Thickness 0.04mm.
Safety
Titanium is a safe material with an excellent history in all the surgical procedures
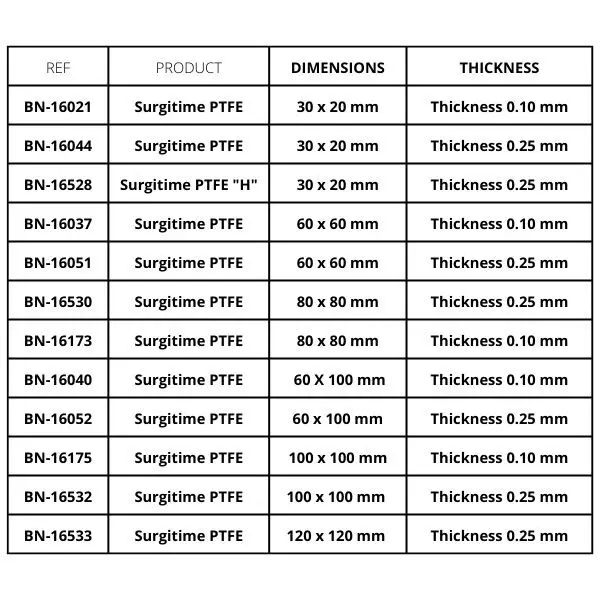
Bionnovation Surgitime PTFE Membrane
Surgitime PTFE membrane Non-absorbable membrane composed of Polytetrafluoroethylene (PFTE).
Features
• High resistance (weight/resistance ratio);
• Chemically inert;
• High chemical resistance in aggressive environments;
• Low inflammability;
• Low coefficient of friction;
• Low dielectric constant;
• Good weathering properties;