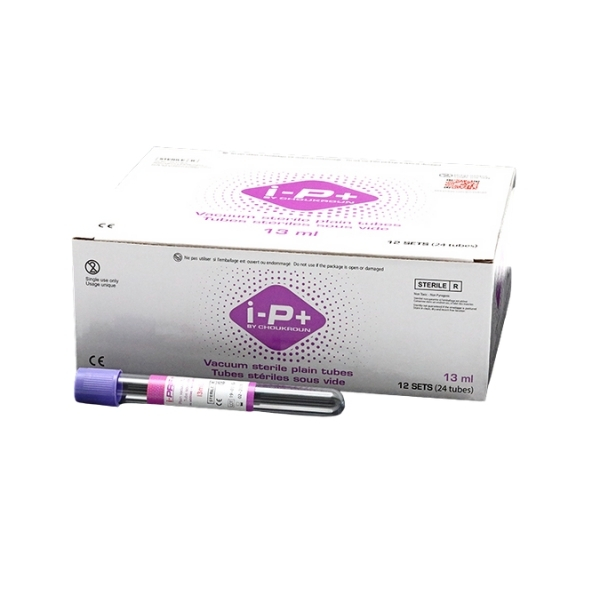
I-PRF Purple Tubes
I-PRF Purple Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Immediate Platelet-Rich Fibrin (I-PRF) from autologous blood. These tubes are designed for clinical use in regenerative medicine and aesthetic procedures.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF). CE 0123 The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF). CE 0123 The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence
Expiry date: 04-2027
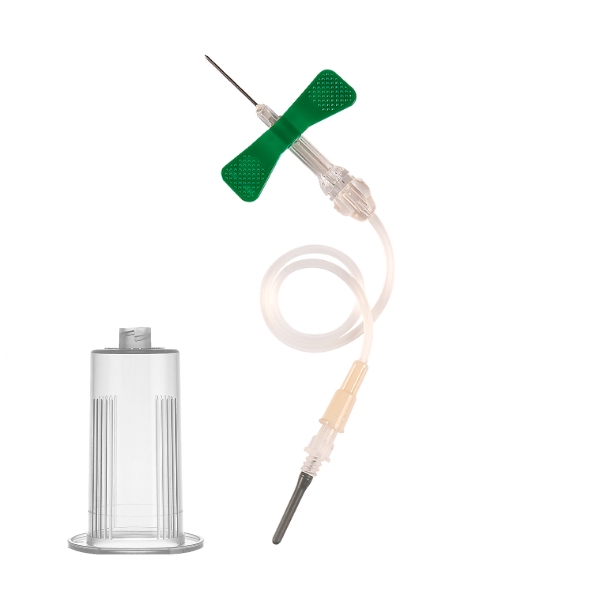
Blood collection butterfly with retractable needle
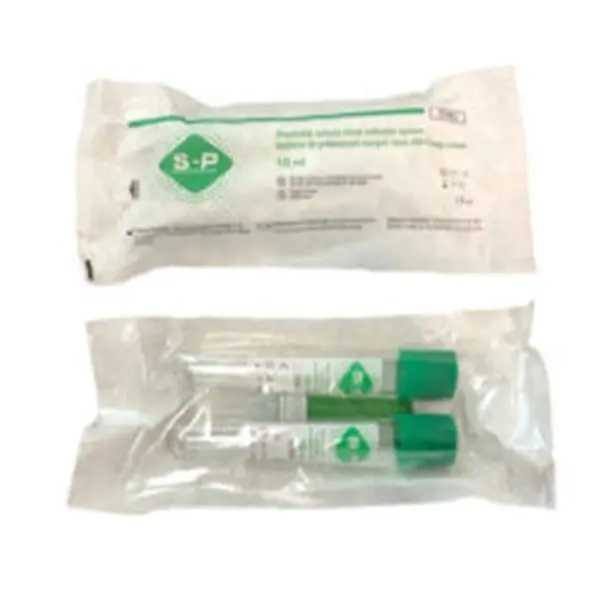
S-PRF Green Tubes
S-PRF Green Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Liquid Platelet-Rich Fibrin (S-PRF) from autologous blood in dental and medical procedures.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF). CE 0123 The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF
See Supporting Research and Clinical Evidence
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF). CE 0123 The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF
See Supporting Research and Clinical Evidence
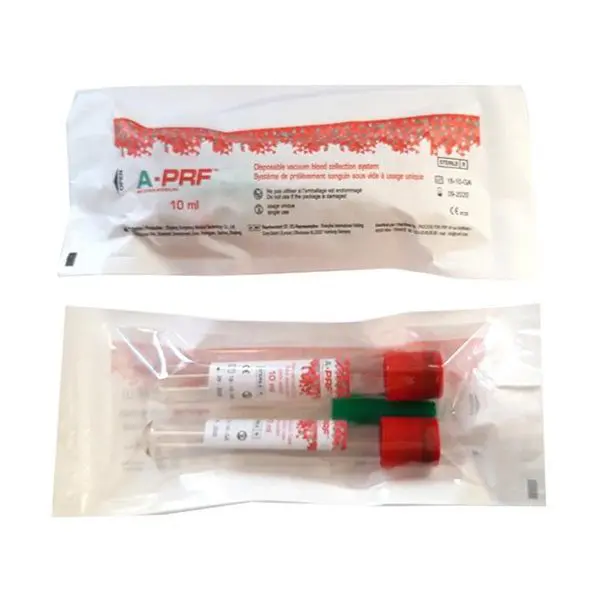
A-PRF Red Tubes
A-PRF Red Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Solid Platelet-Rich Fibrin (A-PRF) from autologous blood. They are specifically designed to facilitate the preparation of PRF clots and membranes for clinical use.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF). CE 0123 The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF). CE 0123 The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence
PRF in Dentistry & Venipuncture Course
PRF in Dentistry (Includes Venipuncture Certification)
Enhance your clinical capabilities with the PRF and Venipuncture Course, specifically designed for dental practitioners. This comprehensive one day intensive program combines evidence based theory with hands on clinical training, focusing on the safe collection, preparation, and application of Platelet Rich Fibrin (PRF) in dentistry.
Dates & Locations:We strongly recommend that dentists bring a staff member to assist with PRF preparation. This allows clinicians to focus fully on venipuncture training and clinical application.
March 28th 2026 | Sydney | 694-696 Elizabeth St, Waterloo, NSW 2017 (SOLD OUT)
August 29th 2026 | Sydney | 694-696 Elizabeth St, Waterloo, NSW 2017
November 28th 2026 | Melbourne - Venue TBC
PRF DUO QUATTRO Centrifuge
The DUO Quattro is an advanced centrifuge designed for the preparation of autologous blood concentrates.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
See Supporting Research and Clinical Evidence
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
See Supporting Research and Clinical Evidence
PRF System Starter
PRF System Starter
Our carefully selected PRF Starter kit is designed for clinicians who are beginning their use of Platelet-Rich Fibrin in oral surgery, implantology, and regenerative dentistry. It provides all the essential components to confidently perform PRF protocols in a safe, reliable, and clinically accurate manner.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
See Supporting Research and Clinical Evidence
The kit also includes complimentary access to Dr. Joseph Choukroun PRF Online Course, valued at $1,000 offering in-depth training from the inventor of Process for PRF
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
See Supporting Research and Clinical Evidence
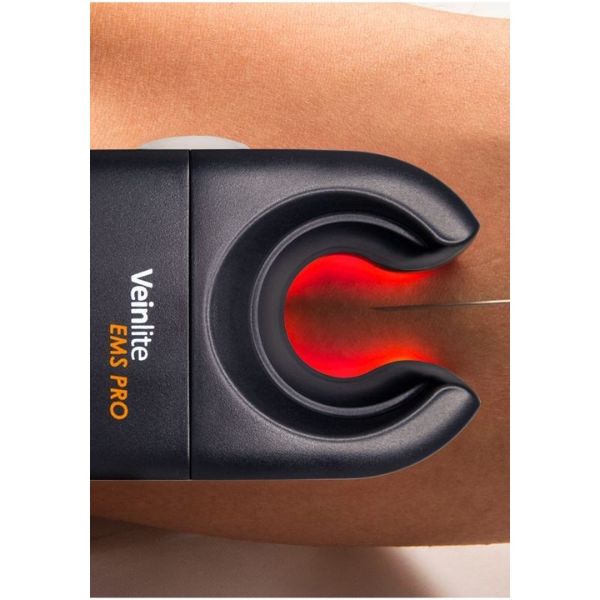
Vein Finder Veinlite EMS PRO
Vein Finder Veinlite EMS PRO
EMS PRO's patented lateral transillumination technology penetrates the skin's surface to reveal the structure of deep and superficial veins.
Veinlite EMS Pro locates the vein and facilitates needle insertion.
Powered by two AA batteries, it features 24 colored LEDs activated by a single switch.
Veinlite EMS Pro eliminates uncertainty in vein location.
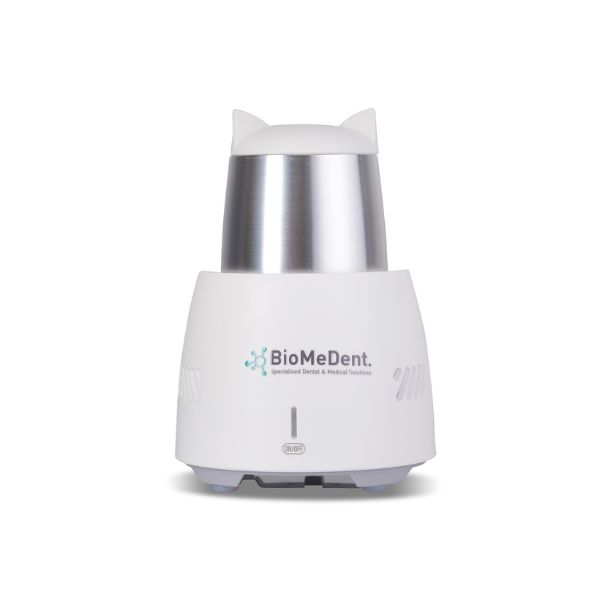
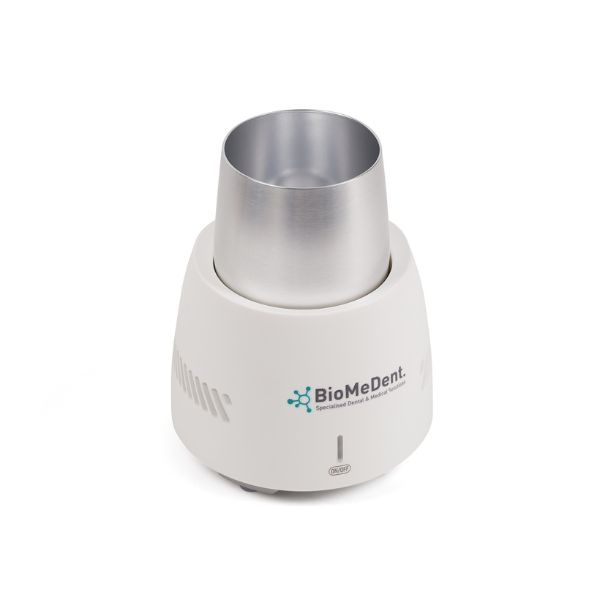
PRF Cooling Device (Cooler)
Device intended for controlled cooling of centrifuged autologous blood components. Designed for professional use in clinical or laboratory settings. Optimizes handling of liquid PRF for subsequent clinical procedures.
Note: Avoid efficacy or percentage improvement claims in patient-facing materials.
See Supporting Research and Clinical Evidence
See Supporting Research and Clinical Evidence