Hide price – login required
Female-Female Luer Lock Syringe Connectors
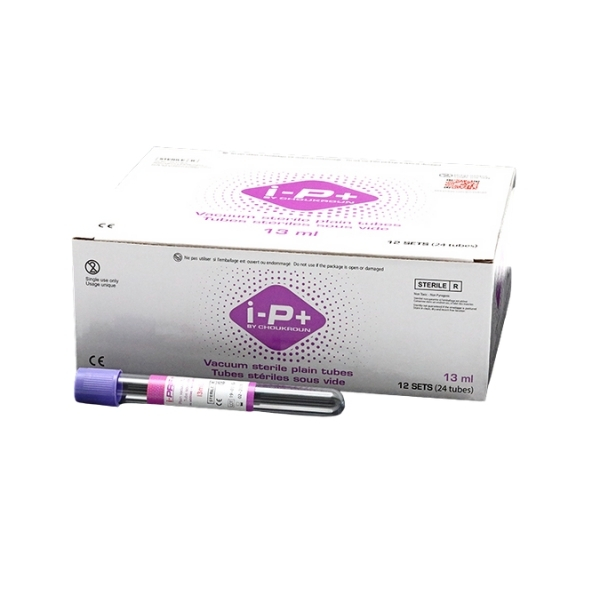
I-PRF Purple Tubes
I-PRF Purple Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Immediate Platelet-Rich Fibrin (I-PRF) from autologous blood. These tubes are designed for clinical use in regenerative medicine and aesthetic procedures.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
CE 0123
The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence
Expiry date: 04-2027
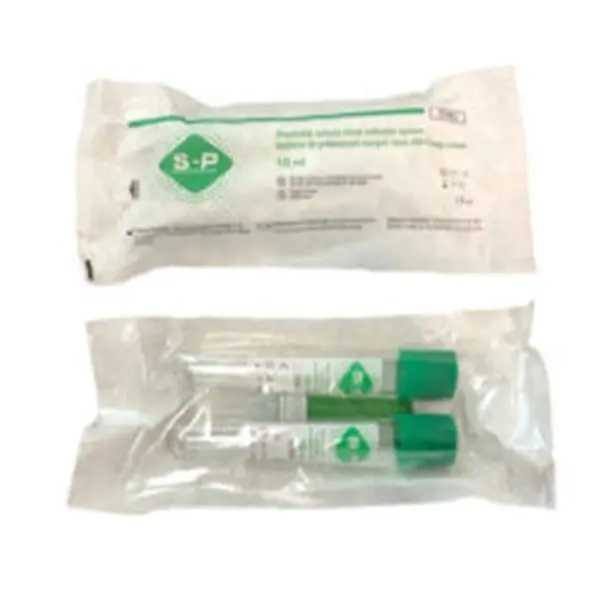
S-PRF Green Tubes
S-PRF Green Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Liquid Platelet-Rich Fibrin (S-PRF) from autologous blood in dental and medical procedures.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
CE 0123
The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF
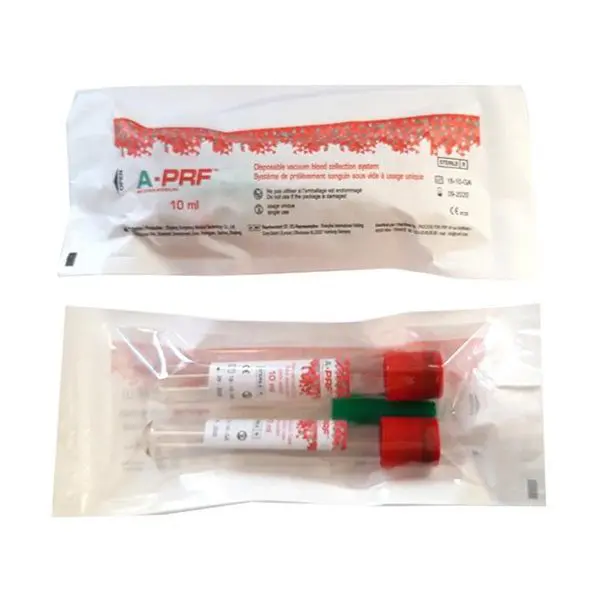
A-PRF Red Tubes
A-PRF Red Tubes are sterile, plain vacuum tubes without additives, anticoagulants, or gels, intended for the preparation of Solid Platelet-Rich Fibrin (A-PRF) from autologous blood. They are specifically designed to facilitate the preparation of PRF clots and membranes for clinical use.
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).
CE 0123
The four-digit number indicates the Notified Body responsible for conformity assessment. Devices without a four-digit CE number are not medical devices and must not be used for PRF.
See Supporting Research and Clinical Evidence
Triple Tipped Luer Lock Needles
Luer Lock Heat Resistant Syringe 1ml
Luer Lock Heat Resistant Syringe 3ml
POMPAC from Process for PRF – Dr Joseph Choukroun
Brand: Process for PRF

The most important innovation in the field of PRF in the last 20 years!
In order to constantly improve the technique and results of PRF, discover our new product : PomPac™
With PomPac™ users will obtain larger PRF clots and liquid PRF with 50% more volume!
https://www.youtube.com/watch?v=0CoeS-p_Tyc
PRF Tube Holder
PRF DUO QUATTRO Centrifuge
PRF System Starter
PRF System Starter
Our carefully selected PRF Starter kit is designed for clinicians who are beginning their use of Platelet-Rich Fibrin in oral surgery, implantology, and regenerative dentistry. It provides all the essential components to confidently perform PRF protocols in a safe, reliable, and clinically accurate manner.
The kit also includes complimentary access to Dr. Joseph Choukroun PRF Online Course, valued at $1,000 offering in-depth training from the inventor of Process for PRF
Regulatory Status:
ARTG-listed Class IIa. Official Process for PRF® product, developed by Dr. Choukroun creator of the Process for PRF® (Platelet Rich Fibrin) technique (A-PRF, i-PRF, & S-PRF).